— This article by Jerry Cates was first published on 11 January 2012, and revised last on 24 October 2015. © Budsinthenews Vol. 3:1(1).
——————————————————-
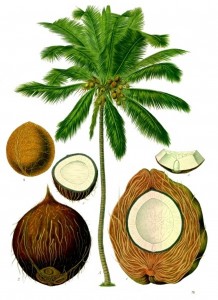
The coconut palm (Cocos nuciferia) is an angiosperm (i.e., a flowering, seed-bearing plant), a monocot (having a single seed leaf), and a member of the Arecaceae (palm) family. It was first described by Carl Heinrich Schultz (1798-1871), the same German botanist who first described the Arecaceae.
The palm family as a whole is recognized, today, as having over 200 genera and about 2600 species. Most are found in tropical, subtropical, and the warmer of earth’s temperate regions, and include a diversity of morphological forms, ranging from vines and shrubs to trees. Fruits vary considerably, from tiny fruits unrecognizable as such to small, soft, sugary dates, and upward in size and heft to the large, heavily encased coconuts depicted by Franz Eugen Köhler in his 1887 book, Medizinal-Pflanzen.
The American botanist Harold Emery Moore, Jr. (1917–1980) devoted much of his career to work on the systematics of the palm family. His research culminated, in the 1970’s, in the organization of the Arecaceae family into fifteen major morphological groups. In 1987 a revision of Moore’s classification, proposed by Natalie W. Uhl and John Dransfield in their beautifully illustrated book, Genera Palmarum: A Classification of Palms Based on the Work of Harold E. Moore, reorganized the palms into six subfamilies, the largest being the Arecoideae, which includes subject of this article, the coconut palm.
The generic name Cocos seems, superficially, to be derived from the Greek word κοκκος, which means “a grain, berry, or kernel.” It happens, though, that the word cocos, which is common to the Spanish and Portuguese languages, has the meaning of “a grinning face,” and it was this name that was applied, early on, to the denuded shell of the coconut fruit. With the covering husk removed to expose the thin, brown, hardened semi-spherical casing surrounding the white endosperm and the liquid coconut water it encloses, the shell has, at its proximal end, three darkened depressions that have the appearance of a human or monkey visage.
The genus Cocos has but one species, nucifera, a specific epithet applied by Schultz, probably by combining two Latin words: nucelus = a kernel, a little fruit + the Latin word ferax = fertile, fruitful, prolific. The name is appropriate. Coconut palms are distributed throughout most of the earth’s the tropical and subtropical regions, where every part of the fruit is utilized for a variety of domestic, commercial, and industrial purposes. The coconut fruit — which is not a true nut but a drupe — consists of an endosperm of white, edible, meat that in dried form is called copra. The oil and milk derived from copra are used in cooking, soap-making, and as an important ingredient in cosmetics.
Liquid coconut water, which is sterile when freshly removed from the coconut fruit’s interior, is a nutritious, refreshing beverage. Though its chemistry is unlike blood plasma in several respects, it is similar enough to make it suitable for use, under emergency conditions, in blood transfusions. WWII reports of such usage are well documented, as are similar reports of native Vietnamese soldiers being so treated on the battlefields of the War in Vietnam.
Coconut husks and leaves are often used as raw materials for the manufacture of furniture, bedding, and for kitchen and eating implements.
Coconut meat is comprised of a high fraction of lipids, or oils. These are divided roughly into nine classes of oils, based on the number of carbon atoms (8-18) in the fatty acid molecule. From 77-89% of these oils are capric (C10H20O2 [or CH3(CH2)8COOH], 6–10%), lauric (C12H24O2, 46–50%), myristic (C14H28O2, 17–19%), and palmitic (C16H32O2, 8–10%) fatty acids (Gervajio, 2005). Each of these fatty acids is more fully described below:
Capric acid:
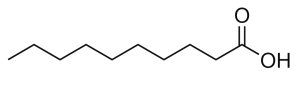
Capric acid, also known as decanoic acid, is a saturated fatty acid with the chemical formula CH3(CH2)8COOH. Its salts and esters are termed decanoates or “caprates”.
Capric acid is a C10 medium-chain saturated fatty acid with the chemical formula CH3(CH2)8COOH and the systematic name n-decanoic acid. It is one of twelve carboxylic acids found in nature. Its salts and esters are referred to as decanoates. The name capric is a derivation of the Latin word capra = “a she-goat,” in reference to the odor of the pure lipid. Two other carboxylic fatty acids, caproic (C6)and caprylic (C8) acid, received their common names via the same etymological route.
This fatty acid comprises 6-10% of natural coconut oil, and 3-5% of palm kernel oil. It is also present in the milks of some mammals and the fats of a number of different animals. It shows considerable influence on animal biology, in a variety of ways. For example, a study published in 1991 reported on the ability of capric acid to relax human blood vessels, an effect with promise in the treatment of stroke victims (White et al. 1991).
Other studies indicate that the presence of capric acid effectively inhibits the development of certain yeasts, including Candida albicans. The latter is the most common opportunistic fungal pathogen isolated from human body, and is well known as the cause of superficial and systemic human diseases (Murzyn et al. 2010).
Of 11 fatty acids and monoglycerides tested against the microbe Campylobacter jejuni, which is one of the most common causes of human gastroenteritis in the world, the 1-monoglyceride of capric acid (monocaprin) was found to be the most effective at killing it. The researchers in that study prepared various monocaprin-in-water emulsions, finding them stable after storage at room temperature for many months during which they retained their microbicidal capabilities (Thormar et al. 2006).
Lauric acid:
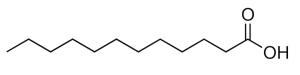
Lauric acid, also known as dodecanoic acid, is a saturated fatty acid with a 12-carbon atom chain, thus falling into the medium chain fatty acids. It is a white, powdery solid with a faint odor of bay oil or soap.
Lauric acid is a C12 medium-chain saturated fatty acid with the chemical formula CH3(CH2)10COOH, and the systematic name dodecanoic acid. It and myristic acid, discussed below, have been the subjects of much speculation, negative and positive, regarding their association with cholesterol in human biology.
Because these medium-chain saturated fatty acids have a melting point above room temperature (solidifying at 76 degrees F. and below), they were initially classed with saturated fatty acids of animal origin — such as the stearic acid in beef tallow and pork lard — that also solidify at room temperature. This led to the supposition that their regular consumption, by humans, would produce similar, if not identical, pathological conditions. The presumption that the fatty acids in coconut oil — particularly the two most prevalent, lauric and myristic acids — are atherogenic (i.e., are agents that cause atherosclerosis), originated from this belief.
Nutritional science moves slowly. If, as many today believe, the supposed atherogenic effect of coconut oil is not only incorrect but off by as much as 180 degrees (i.e., instead of causing atherosclerosis, coconut oil actually reduces the risk of atherogenesis, making coconut oil what some call a miracle oil), it will take time and a number of rigorous, convincing, scientific studies to bring the point home. Fortunately, the scientific community has risen to the challenge, and a host of studies, some spanning several decades in time, have sought answers to these very questions. Some are listed below, under references to scientific papers (all references are linked to Internet sources where the paper may be read in full), and interested persons are encouraged to click on the link and read through them.
Scientific analysis suggests that while coconut oil is not the villain it is often portrayed as, it is also not the miracle oil its protractors claim it to be. The various lipids contained in coconut oil behave in unique ways within the human body, and the differences between them significantly affect their usefulness in human nutrition as well as in all the other ways these oils are used in our daily lives.
Myristic acid:
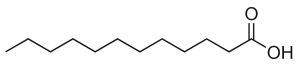
Myristic acid, also known as tetradecanoic acid, is a common saturated fatty acid with the molecular formula CH3(CH2)12COOH. A myristate is a salt or ester of myristic acid. Myristic acid is named after the nutmeg Myristica fragrans.
Myristic acid is a C14 medium-chain saturated fatty acid with the chemical formula CH3(CH2)12COOH, and the systematic name tetradecanoic acid. The name is derived from its most concentrated source, the nutmeg, Myristica fragrans. Nutmeg butter is comprised of 75% trimyristin, the triglyceride form of myristic acid.
Myristic acid is a major constituent of palm kernel oil, coconut oil, and butter fat, and is also present in small amounts in a number of animal fats, including spermacetin, the crystallized fraction of oil from the sperm whale. Myristic Acid is often used as a cleansing agent, a surfactant, and an opacifier, in a variety of products, including cosmetics and personal care formulas.
Palmitic acid:
![Palmitic acid, also known as hexadecanoic acid, is the most common fatty acid (saturated) found in animals, plants and microorganisms.[9] Its chemical formula is CH3(CH2)14COOH. As its name indicates, it is a major component of the oil from palm trees (palm oil), but can also be found in meats, cheeses, butter, and dairy products.](https://budsinthenews.info/wp-content/uploads/2012/01/Palmitic-acid-300x54.png)
Palmitic acid, also known as hexadecanoic acid, is the most common fatty acid (saturated) found in animals, plants and microorganisms.[9] Its chemical formula is CH3(CH2)14COOH. As its name indicates, it is a major component of the oil from palm trees (palm oil), but can also be found in meats, cheeses, butter, and dairy products.
Palmitic acid is the first fatty acid produced during fatty acid synthesis (Excess carbohydrates in the body are converted to palmitic acid) and the precursor to longer fatty acids. As a consequence, palmitic acid is a major body component of animals. In humans, one analysis found it to comprise 21–30% (molar) of human depot fat, and it is a major, but highly variable, lipid component of human breast milk. Palmitate negatively feeds back on acetyl-CoA carboxylase (ACC), which is responsible for converting acetyl-CoA to malonyl-CoA, which in turn is used to add to the growing acyl chain, thus preventing further palmitate generation. In biology, some proteins are modified by the addition of a palmitoyl group in a process known as palmitoylation. Palmitoylation is important for membrane localisation of many proteins.
Palmitic acid is used to produce soaps, cosmetics, and release agents. These applications utilize sodium palmitate, which is commonly obtained by saponification of palm oil. To this end, palm oil, rendered from palm tree (species Elaeis Guineensis), is treated with sodium hydroxide (in the form of caustic soda or lye), which causes hydrolysis of the ester groups. This procedure results in the production of glycerol and sodium palmitate.
Because it is inexpensive and adds texture to processed foods (convenience food), palmitic acid and its sodium salt find wide use including foodstuffs. Sodium palmitate is permitted as a natural additive in organic products. The aluminum salt has been used as a thickening agent in the production of napalm used in military actions.
Hydrogenation of palmitic acid yields cetyl alcohol, which is used to produce detergents and cosmetics.
Recently, a long-acting antipsychotic medication, paliperidone palmitate (marketed as INVEGA Sustenna), used in the treatment of schizophrenia, has been synthesized using the oily palmitate ester as a long-acting release carrier medium when injected intramuscularly. The underlying method of drug delivery is similar to that used with decanoic acid to deliver long-acting depot medication, in particular, neuroleptics such as haloperidol decanoate.
—————————————–
Taxonomy:
- Domain: Eukaryota (yoo-carr-ee-OH-tah) — from the Greek prefix ευ (yew) = good, well, pleasing + καρυον (khar-yone) = a nut/nucleus, thus organisms whose cells contain a nucleus and other organelles within membranes.
- (unranked): Bikonta Cavalier-Smith, 1993 (by-KOHN-tuh) — from the Latin bis = twice/double + the Greek κοντος = a punting pole; those eukaryotic organisms within the subgroups Apusozoa, Rhizaria, Excavata, Archaeplastida, or Chromalveolata.
- (unranked): Archaeplastida Adl et al., 2005 (ahr-kee-PLASS-tih-duh) — from the Greek αρχαιος (AHR-kee-ose) = ancient/antiquated + πλασις (PLAS-iss) = a moulding + Anglo Saxon tid = time; a major group of eukaryotes, comprised of the red algae (Rhodophyta), the green algae, and the land plants along with the freshwater unicellular algae known as glaucophytes.
- Kingdom/Regnum: Plantae Copeland, 1956 (PLAN-tee) or Viridiplantae Cavalier-Smith, 1881 (veer-id-eye-PLAN-tee) — from the Latin planta = a green twig; the plant kingdom, consisting of multi-cellular green plants, i.e., whose cells have cellulose within their cell walls and have primary chloroplasts derived from endosymbiosis with cyanobacteria containing chlorophylls a and b and lack phycobilins..
- (unranked): Streptophyta Jeffrey 1967 (strepp-toh-PHY-tah) — from στρεπτος (STREP-tose) = (easily) twisted, pliant + φυτον (PHU-tawn) = a plant/tree; the land plants and the green algal group Charophyta.
- Subkingdom: Embryophyta Engler, 1892 (imm-bree-oh-FYE-tuh) — from the Greek εμβρυον (EMM-bree-yon) + φυτον (PHU-tawn) = a plant/tree; green plants, informally known as land plants because most are terrestrial rather than aquatic, while the related green algae are primarily aquatic;
- (unranked): Angiosperms (AN-gee-oh-spurms)/Magnoliophyta Cronquist (mag-NOH-lee-oh-fye-tuh) — from the Greek αγγειον (AUGG-ee-awn) = a vessel/pail/reservoir + σπερμα (SPUR-mah) = a seed; the flowering plants, distinguished from the gymnosperms by having flowers, endosperm within the seeds, and the production of fruits that contain the seeds;
- (unranked) Monocots (MONN-oh-kotts) — from the Greek μοηας (MOH-nus) = single/solitary/a unit + κοτυληδων (cott-ee-LEE-dun) = a cup-shaped hollow; a clade within the angiosperms
- (unranked) Commelinids (kumm-ehl-LYNN-iddz) — a named derived from a genus (Commelina) named by the Swedish taxonomist Carl Linnaeus in honor of the Dutch botanist Jan Commelijin; a clade within the monocots
- Order: Arecales (ahr-ee-KAHL-ees) — a monocot order comprised of a single family, Arecaceae, the palm trees, with roughly 200 genera and 2600 species;
- Family: Arecaceae (ahr-ee-KAY-say-ee) — palm trees; comprised of five subfamilies and sixteen tribes.
- Subfamily: Arecoideae (arr-ee-KOY-day-ee) — a subfamily of the palm tree family, comprised of six tribes;
- Tribe: Cocoeae (koh-koh-AY-ee) — one of the six tribes recognized within the Arecoideae subfamily of palm trees, comprised of 18 genera: 1. Acrocomia (spiny, pinnate-leaved palms; 8 recognized species; 2. Aiphanes (spiny palms; 26 recognized species); 3. Allagoptera (monoecious flowering palms with very short or acaulescent trunks that tend to bifurcate, producing multiple heads; 5 recognized species); 4. Astrocaryum (spiny, pleonanthic, monoecious palms with pinnately compound leaves, comprised of 36-40 recognized species); 5. Attalea (pinnately leaved, non-spiny palms distinguished by their male flowers; comprised of 29-67 recognized species); 6. Bactris (a highly speciated genus of spiny, mostly small palms with simple or pinnately compound leaves and yellow, orange, red, or purple-black fruit, currently having 79 recognized species), ; 7. Barcella (a monotypic genus comprising a single species of flowering palm, Barcella odora, which is used by Brazilian peoples in the states of Amazonas and Roraima for construction and thatched goods); 8. Beccariophoenix (closely related to the genus Cocos, and similar in appearance to the single species of that genus; three recognized species); 9. Butia (feather palms with pinnate leaves; comprised of 22 recognized species); 10. Cocos (comprised of a single species, Cocos nucifera, described more fully below); 11. Desmoncus (spiny palms native to the Neotropics; comprised of 24 recognized species); 12. Elaeis (a genus of two species of oil palms); 13. Jubaea (a genus of one extant species, Jubaea chilensis, the Chilean wine palm, a slow growing tree whose sap has been used to produce a fermented beverage); 14. Jubaeopsis (a monotypic genus comprised of the single species Jubaeopsis caffra, a flowering palm found only in South Africa, a relic of a large lineage of palm trees widespread in southern Africa in prehistoric times); 15. Lytocaryum (monoecious flowering palms endemic to Brazil comprised of 4 recognized species); 16. Parajubaea (a genus of 3 species of flowering palms native to the northern Andes mountains in northwestern South America); 17. Syagrus (a genus of palms closely related to the genus Cocos, with edible seeds similar to that of the coconut; comprised of 61 recognized species); 18. Voanioala (a monotypic palm known as the forest coconut, endemic to Madagascar, and now extremely rare; estimated to be represented today by no more than 15 trees);
- Genus: Cocos L. (KOH-kohs) — coconut palm trees.
- Species: C. nucifera (new-SIFF-err-ah) L. — the coconut palm tree, the only species within the genus Cocos.
—————————————–
References to Scientific Papers:
- Awua, Adolf K., et al. 2011. Exploring the influence of sterilisation and storage on some physicochemical properties of coconut (Cocos nucifera L.) water. BMC Research Notes 2011, 4:451.
- Connor, William E. 1999. Harbingers of coronary heart disease: dietary saturated fatty acids and cholesterol. Is chocolate benign because of its stearic acid content? Am J Clin Nutr 1999;70:951–2.
- Dabadie, H., et al. 2005. Moderate intake of myristic acid in sn-2 position has beneficial lipidic effects and enhances DHA of cholesteryl esters in an interventional study. J Nutr Biochem. 16(6):375-82.
- de Roos, Nicole M., et al. 2001. Consumption of a Solid Fat Rich in Lauric Acid Results in a More Favorable Serum Lipid Profile in Healthy Men and Women than Consumption of a Solid Fat Rich in trans-Fatty Acids. J. Nutr. 131: 242–245.
- Erguiza, G. S., et al. 2008. The effect of virgin coconut oil supplementation for community-acquired pneumonia in children aged 3 to 60 months admitted at the Philippine Children’s Medical Center: a single blinded randomized controlled trial. Chest. 2008;134:139001.
- Gervajio, Gregorio C. 2005. Fatty Acids and Derivatives from Coconut Oil. Bailey’s Industrial Oil and Fat Products, Sixth Ed., John Wiley & Sons, Inc.
- Hu, Frank B. et al. 1999. Dietary saturated fats and their food sources in relation to the risk of coronary heart disease in women. Am J Clin Nutr 1999;70:1001–8.
- Lieberman, Shari, et al. 2006. A Review of Monolaurin and Lauric Acid: Natural Virucidal and Bactericidal Agents. Alternative & Complementary Therapies, Dec. 2006:310-314.
- Lipoeto, Nur I., et al. 2001. Contemporary Minangkabau food culture in West Sumatra, Indonesia. Asia Pac J Clin Nutr. 10:10-6.
- Lipoeto, Nur I., et al. 2004. Dietary intake and the risk of coronary heart disease among the coconut-consuming Minangkabau in West Sumatra, Indonesia. Asia Pac J Clin Nutr 13 (4):377-384
- Mensink, Ronald P., et al. 2003. Effects of dietary fatty acids and carbohydrates on the ratio of serum total to HDL cholesterol and on serum lipids and apolipoproteins: a meta-analysis of 60 controlled trials. Am J Clin Nutr. 77:1146-55.
- Murzyn, Anna, et al, 2010. Capric Acid Secreted by S. boulardii Inhibits C. albicans Filamentous Growth, Adhesion and Biofilm Formation. PLoS ONE 5(8): e12050.
- Nakatsuji, Teruaki, et al. 2009. Antimicrobial Property of Lauric Acid Against Propionibacterium acnes: Its Therapeutic Potential for Inflammatory Acne Vulgaris. J Invest Dermatol. 129(10): 2480–2488.
- Nevin, K. G., and T. Rajamohan. 2004. Beneficial effects of virgin coconut oil on lipid parameters and in vitro LDL oxidation. Clin Biochem 37:830-5.
- Ng, Tony K. W. et al. 1991. Nonhypercholesterolemic effects of a palm-oil diet in Malaysian volunteers. Am JClin Nutr 53:1015-20.
- Oh, Kyungwon, et al. 2004. Dietary Fat Intake and Risk of Coronary Heart Disease in Women: 20 Years of Follow-up of the Nurses’ Health Study. Am J Epidemiol 2005;161:672–679
- Pattigadapa, Hemanth Sairam, et al. 2011. Cardiotonic Activity of Coconut Water (Cocos nucifera). Recent Research in Science and Technology 3(4):155-157.
- Prior, Ian A., et al. 1981. Cholesterol, coconuts, and diet on Polynesian atolls: a natural experiment: the Pukapuka and Tokelau Island studies. Am. J. Clin. Nutr. 34: 1552-1561.
- Sengupta A, and M. Ghosh. 2011. Comparison of native and capric acid-enriched mustard oil effects on oxidative stress and antioxidant protection in rats. Br J Nutr. 2011 Nov 1:1-5.
- Solangi, A. H., and M. Z. Iqbal. 2011. Chemical Composition of Meat (Kernel) and Nut Water of major Coconut (Cocos nucifera L.) Cultivars at Coastal Area of Pakistan. Pak. J. Bot., 43(1): 357-363.
- Thormar, Haldor, et al. 2006. Stable Concentrated Emulsions of the 1-Monoglyceride of Capric Acid (Monocaprin) with Microbicidal Activities against the Food-Borne Bacteria Campylobacter jejuni, Salmonella spp., and Escherichia coli. Applied and Environmental Microbiology, 72 (Jan 2006):522-526.
- Ugbogu, O. C., et al. 2006. Short Communication: Lauric acid content and inhibitory effect of palm kernel oil on two bacterial isolates and Candida albicans. African Journal of Biotechnology Vol. 5 (11), pp. 1045-1047.
- Uhl, Natalie W. and John Dransfield. 1987. Genera Palmarum: A Classification of Palms Based on the Work of Harold E. Moore, Jr. Publ. by Genera Palmarum.
- White, Richard P., et al. 1991. Identification of Capric Acid as a Potent Vasoreiaxant of Human Basilar Arteries. Stroke, 22(4):469-476.
- Yong, Jean W. H. et al. 2009. The Chemical Composition and Biological Properties of Coconut (Cocos nucifera L.) Water. Molecules 2009, 14, 5144-5164.